The U.S. government on Thursday will lay out its long-awaited plan to ban menthol cigarettes and flavored cigars, which have taken a disproportionate toll on Black smokers and other minorities.
Food and Drug Administration Commissioner Robert Califf previewed the announcement in congressional testimony, saying the proposal would reduce disease and death by helping current smokers quit and stopping younger people from starting.
The agency has been under legal pressure to issue a decision after anti-smoking and civil rights groups sued the FDA for “unreasonably” delaying action on earlier requests to ban menthol. Menthol’s cooling effect has been shown to mask the throat harshness of smoking, making it easier to start and harder to quit smoking.
Menthol is the only cigarette flavor that was not prohibited under the 2009 law that gave the FDA authority over tobacco products, an exemption negotiated by industry lobbyists. The act did, though, instruct the agency to continue to weigh a ban.within the year, responding in part to African American groups who say menthol has led to lower quit rates and higher rates of death among Black people. Menthols are used by 85% of Black smokers.
Österreich Neuesten Nachrichten, Österreich Schlagzeilen
Similar News:Sie können auch ähnliche Nachrichten wie diese lesen, die wir aus anderen Nachrichtenquellen gesammelt haben.
 FDA Announces Recall of Popular Blood Pressure Medication Due to Potential Cancer RiskIt’s time to double-check your medicine cabinets.
FDA Announces Recall of Popular Blood Pressure Medication Due to Potential Cancer RiskIt’s time to double-check your medicine cabinets.
Weiterlesen »
 FDA Approves First COVID Treatment for Use in KidsThe FDA has approved the antiviral remdesivir as the first COVID-19 treatment for young children.
FDA Approves First COVID Treatment for Use in KidsThe FDA has approved the antiviral remdesivir as the first COVID-19 treatment for young children.
Weiterlesen »
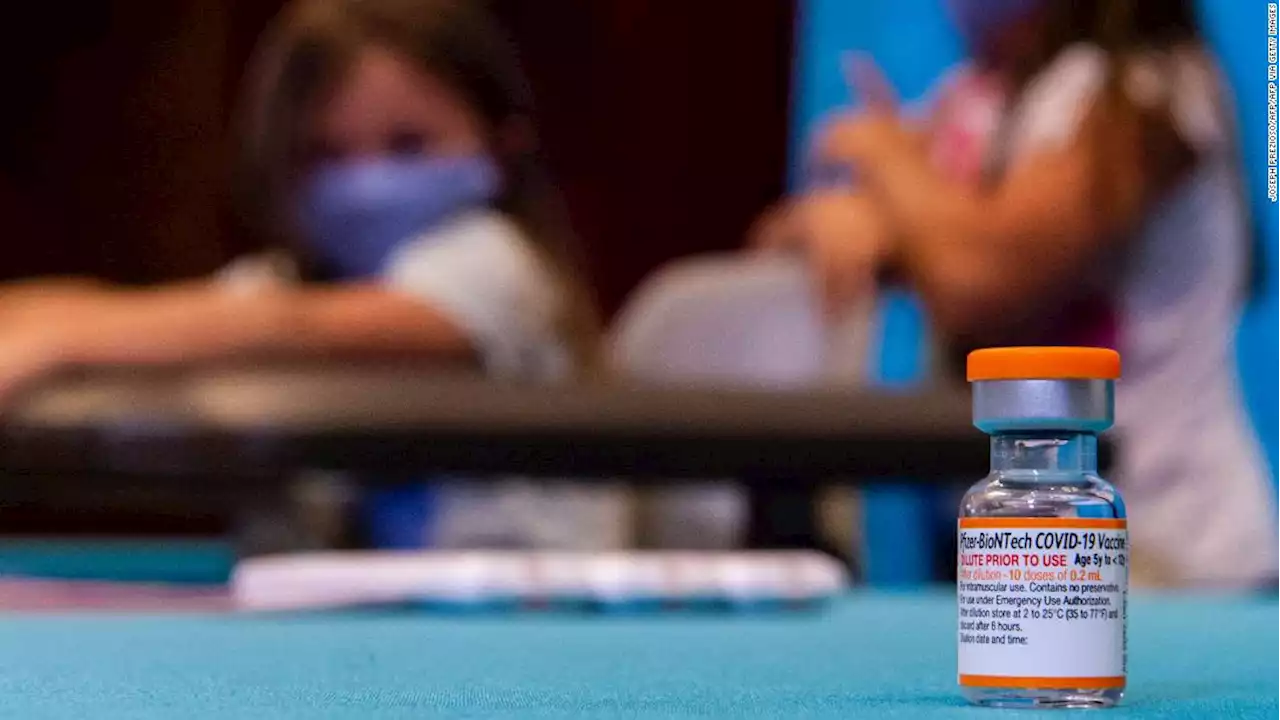 Pfizer requests FDA authorization for Covid-19 booster for kids 5 through 11Pfizer and BioNTech said Tuesday that they have submitted an application to the US Food and Drug Administration for emergency use authorization of a booster dose of Covid-19 vaccine for children 5 through 11.
Pfizer requests FDA authorization for Covid-19 booster for kids 5 through 11Pfizer and BioNTech said Tuesday that they have submitted an application to the US Food and Drug Administration for emergency use authorization of a booster dose of Covid-19 vaccine for children 5 through 11.
Weiterlesen »
 Pfizer Asks FDA to Authorize Third Dose for Children 5 to 11 Years OldPfizer on Tuesday asked the Food and Drug Administration to authorize a third dose of its COVID vaccine for children ages 5- to 11-years-old. The application comes after Pfizer released data earlier this month from a small lab study of blood samples from 30 kids in the age group, which showed a 36-fold increase in antibody levels against the omicron variant…
Pfizer Asks FDA to Authorize Third Dose for Children 5 to 11 Years OldPfizer on Tuesday asked the Food and Drug Administration to authorize a third dose of its COVID vaccine for children ages 5- to 11-years-old. The application comes after Pfizer released data earlier this month from a small lab study of blood samples from 30 kids in the age group, which showed a 36-fold increase in antibody levels against the omicron variant…
Weiterlesen »
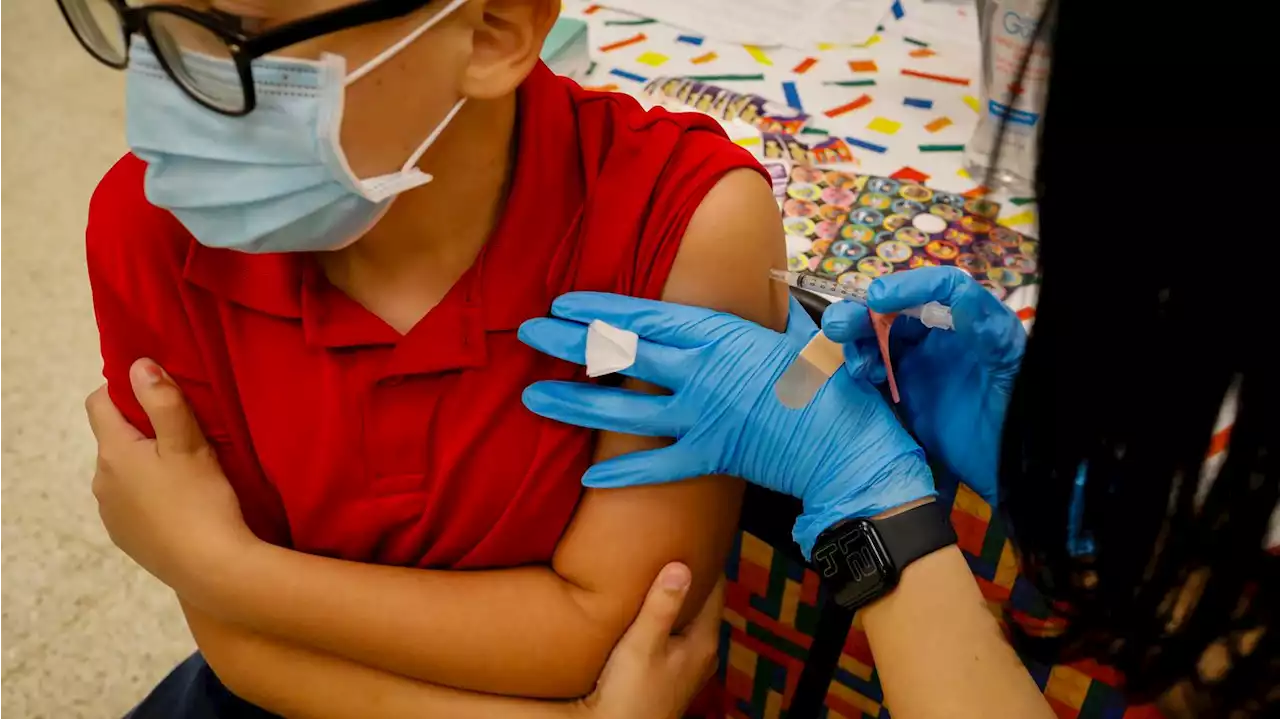 Pfizer asks FDA to authorize COVID booster for children 5 to 11If approved, it would be the first booster accessible to children of this age group, who were hit hard by Omicron.
Pfizer asks FDA to authorize COVID booster for children 5 to 11If approved, it would be the first booster accessible to children of this age group, who were hit hard by Omicron.
Weiterlesen »
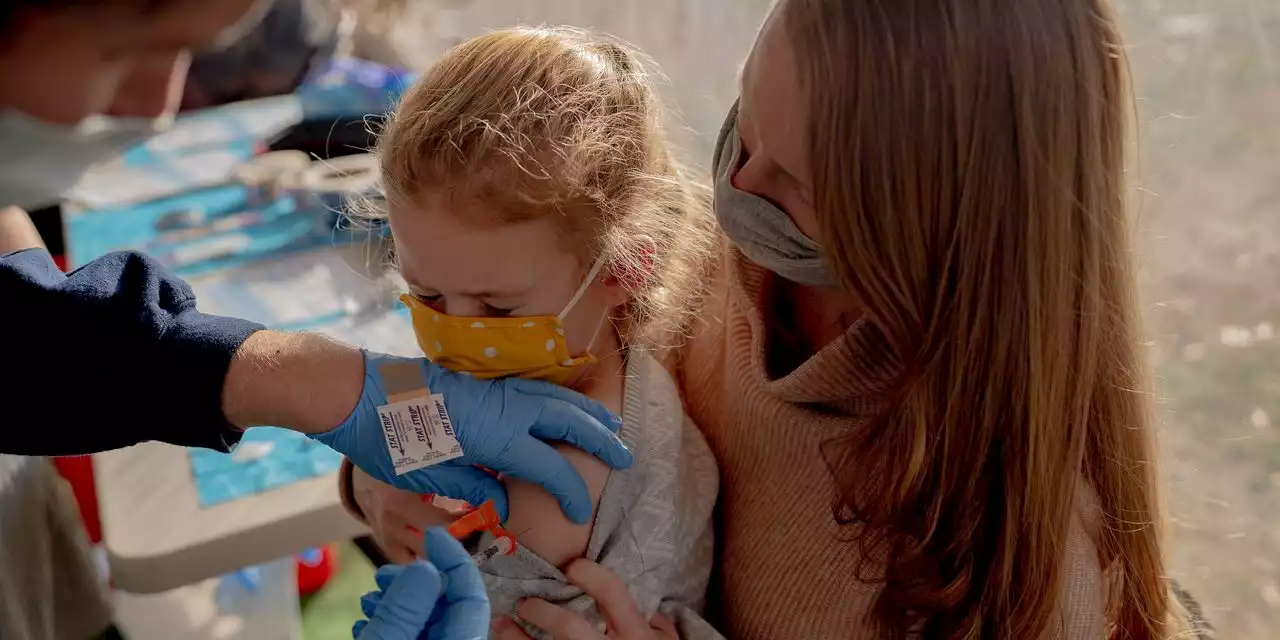 Pfizer Asks FDA to Authorize Covid-19 Booster for 5- to 11-Year-OldsPfizer Inc. and partner BioNTech SE said earlier this month that a third shot safely generated a strong immune response in the youngsters.
Pfizer Asks FDA to Authorize Covid-19 Booster for 5- to 11-Year-OldsPfizer Inc. and partner BioNTech SE said earlier this month that a third shot safely generated a strong immune response in the youngsters.
Weiterlesen »
